List of Tables
Table 1. Global Gene Cell Therapy CDMO Market Size Growth Rate by Type (US$ Million): 2020 VS 2024 VS 2031
Table 2. Key Players of Immune Cells
Table 3. Key Players of Stem Cell
Table 4. Key Players of Viral Vector
Table 5. Key Players of Plasmid DNA
Table 6. Global Gene Cell Therapy CDMO Market Size Growth Rate by Stage (US$ Million): 2020 VS 2024 VS 2031
Table 7. Key Players of Early-stage CDMO and preclinical CDMO
Table 8. Key Players of Clinical-stage CDMO
Table 9. Key Players of Commercial-stage CDMO
Table 10. Global Gene Cell Therapy CDMO Market Size Growth Rate by Production Mode (US$ Million): 2020 VS 2024 VS 2031
Table 11. Key Players of Centralized Factory Model
Table 12. Key Players of Distributed Regional Production Model
Table 13. Global Gene Cell Therapy CDMO Market Size Growth by Application (US$ Million): 2020 VS 2024 VS 2031
Table 14. Global Gene Cell Therapy CDMO Market Size by Region (US$ Million): 2020 VS 2024 VS 2031
Table 15. Global Gene Cell Therapy CDMO Market Size by Region (2020-2025) & (US$ Million)
Table 16. Global Gene Cell Therapy CDMO Market Share by Region (2020-2025)
Table 17. Global Gene Cell Therapy CDMO Forecasted Market Size by Region (2026-2031) & (US$ Million)
Table 18. Global Gene Cell Therapy CDMO Market Share by Region (2026-2031)
Table 19. Gene Cell Therapy CDMO Market Trends
Table 20. Gene Cell Therapy CDMO Market Drivers
Table 21. Gene Cell Therapy CDMO Market Challenges
Table 22. Gene Cell Therapy CDMO Market Restraints
Table 23. Global Gene Cell Therapy CDMO Revenue by Players (2020-2025) & (US$ Million)
Table 24. Global Gene Cell Therapy CDMO Market Share by Players (2020-2025)
Table 25. Global Top Gene Cell Therapy CDMO Players by Company Type (Tier 1, Tier 2, and Tier 3) & (based on the Revenue in Gene Cell Therapy CDMO as of 2024)
Table 26. Ranking of Global Top Gene Cell Therapy CDMO Companies by Revenue (US$ Million) in 2024
Table 27. Global 5 Largest Players Market Share by Gene Cell Therapy CDMO Revenue (CR5 and HHI) & (2020-2025)
Table 28. Global Key Players of Gene Cell Therapy CDMO, Headquarters and Area Served
Table 29. Global Key Players of Gene Cell Therapy CDMO, Product and Application
Table 30. Global Key Players of Gene Cell Therapy CDMO, Date of Enter into This Industry
Table 31. Mergers & Acquisitions, Expansion Plans
Table 32. Global Gene Cell Therapy CDMO Market Size by Type (2020-2025) & (US$ Million)
Table 33. Global Gene Cell Therapy CDMO Revenue Market Share by Type (2020-2025)
Table 34. Global Gene Cell Therapy CDMO Forecasted Market Size by Type (2026-2031) & (US$ Million)
Table 35. Global Gene Cell Therapy CDMO Revenue Market Share by Type (2026-2031)
Table 36. Global Gene Cell Therapy CDMO Market Size by Application (2020-2025) & (US$ Million)
Table 37. Global Gene Cell Therapy CDMO Revenue Market Share by Application (2020-2025)
Table 38. Global Gene Cell Therapy CDMO Forecasted Market Size by Application (2026-2031) & (US$ Million)
Table 39. Global Gene Cell Therapy CDMO Revenue Market Share by Application (2026-2031)
Table 40. North America Gene Cell Therapy CDMO Market Size Growth Rate by Country (US$ Million): 2020 VS 2024 VS 2031
Table 41. North America Gene Cell Therapy CDMO Market Size by Country (2020-2025) & (US$ Million)
Table 42. North America Gene Cell Therapy CDMO Market Size by Country (2026-2031) & (US$ Million)
Table 43. Europe Gene Cell Therapy CDMO Market Size Growth Rate by Country (US$ Million): 2020 VS 2024 VS 2031
Table 44. Europe Gene Cell Therapy CDMO Market Size by Country (2020-2025) & (US$ Million)
Table 45. Europe Gene Cell Therapy CDMO Market Size by Country (2026-2031) & (US$ Million)
Table 46. Asia-Pacific Gene Cell Therapy CDMO Market Size Growth Rate by Region (US$ Million): 2020 VS 2024 VS 2031
Table 47. Asia-Pacific Gene Cell Therapy CDMO Market Size by Region (2020-2025) & (US$ Million)
Table 48. Asia-Pacific Gene Cell Therapy CDMO Market Size by Region (2026-2031) & (US$ Million)
Table 49. Latin America Gene Cell Therapy CDMO Market Size Growth Rate by Country (US$ Million): 2020 VS 2024 VS 2031
Table 50. Latin America Gene Cell Therapy CDMO Market Size by Country (2020-2025) & (US$ Million)
Table 51. Latin America Gene Cell Therapy CDMO Market Size by Country (2026-2031) & (US$ Million)
Table 52. Middle East & Africa Gene Cell Therapy CDMO Market Size Growth Rate by Country (US$ Million): 2020 VS 2024 VS 2031
Table 53. Middle East & Africa Gene Cell Therapy CDMO Market Size by Country (2020-2025) & (US$ Million)
Table 54. Middle East & Africa Gene Cell Therapy CDMO Market Size by Country (2026-2031) & (US$ Million)
Table 55. Catalent Company Details
Table 56. Catalent Business Overview
Table 57. Catalent Gene Cell Therapy CDMO Product
Table 58. Catalent Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 59. Catalent Recent Development
Table 60. Lonza Company Details
Table 61. Lonza Business Overview
Table 62. Lonza Gene Cell Therapy CDMO Product
Table 63. Lonza Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 64. Lonza Recent Development
Table 65. Charles River Company Details
Table 66. Charles River Business Overview
Table 67. Charles River Gene Cell Therapy CDMO Product
Table 68. Charles River Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 69. Charles River Recent Development
Table 70. Oxford Biomedica Company Details
Table 71. Oxford Biomedica Business Overview
Table 72. Oxford Biomedica Gene Cell Therapy CDMO Product
Table 73. Oxford Biomedica Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 74. Oxford Biomedica Recent Development
Table 75. Novartis Company Details
Table 76. Novartis Business Overview
Table 77. Novartis Gene Cell Therapy CDMO Product
Table 78. Novartis Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 79. Novartis Recent Development
Table 80. OBiO Company Details
Table 81. OBiO Business Overview
Table 82. OBiO Gene Cell Therapy CDMO Product
Table 83. OBiO Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 84. OBiO Recent Development
Table 85. GenScript Company Details
Table 86. GenScript Business Overview
Table 87. GenScript Gene Cell Therapy CDMO Product
Table 88. GenScript Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 89. GenScript Recent Development
Table 90. Pharmaron Company Details
Table 91. Pharmaron Business Overview
Table 92. Pharmaron Gene Cell Therapy CDMO Product
Table 93. Pharmaron Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 94. Pharmaron Recent Development
Table 95. Porton Company Details
Table 96. Porton Business Overview
Table 97. Porton Gene Cell Therapy CDMO Product
Table 98. Porton Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 99. Porton Recent Development
Table 100. Resilience Company Details
Table 101. Resilience Business Overview
Table 102. Resilience Gene Cell Therapy CDMO Product
Table 103. Resilience Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 104. Resilience Recent Development
Table 105. SK Pharmteco Company Details
Table 106. SK Pharmteco Business Overview
Table 107. SK Pharmteco Gene Cell Therapy CDMO Product
Table 108. SK Pharmteco Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 109. SK Pharmteco Recent Development
Table 110. Forge Biologics Company Details
Table 111. Forge Biologics Business Overview
Table 112. Forge Biologics Gene Cell Therapy CDMO Product
Table 113. Forge Biologics Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 114. Forge Biologics Recent Development
Table 115. Andelyn Biosciences Company Details
Table 116. Andelyn Biosciences Business Overview
Table 117. Andelyn Biosciences Gene Cell Therapy CDMO Product
Table 118. Andelyn Biosciences Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 119. Andelyn Biosciences Recent Development
Table 120. Roslin CT Company Details
Table 121. Roslin CT Business Overview
Table 122. Roslin CT Gene Cell Therapy CDMO Product
Table 123. Roslin CT Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 124. Roslin CT Recent Development
Table 125. eXmoor Pharma Company Details
Table 126. eXmoor Pharma Business Overview
Table 127. eXmoor Pharma Gene Cell Therapy CDMO Product
Table 128. eXmoor Pharma Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 129. eXmoor Pharma Recent Development
Table 130. Advanced Therapies Company Details
Table 131. Advanced Therapies Business Overview
Table 132. Advanced Therapies Gene Cell Therapy CDMO Product
Table 133. Advanced Therapies Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 134. Advanced Therapies Recent Development
Table 135. Catalent Biologics Company Details
Table 136. Catalent Biologics Business Overview
Table 137. Catalent Biologics Gene Cell Therapy CDMO Product
Table 138. Catalent Biologics Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 139. Catalent Biologics Recent Development
Table 140. Patheon Pharma Services Company Details
Table 141. Patheon Pharma Services Business Overview
Table 142. Patheon Pharma Services Gene Cell Therapy CDMO Product
Table 143. Patheon Pharma Services Revenue in Gene Cell Therapy CDMO Business (2020-2025) & (US$ Million)
Table 144. Patheon Pharma Services Recent Development
Table 145. Research Programs/Design for This Report
Table 146. Key Data Information from Secondary Sources
Table 147. Key Data Information from Primary Sources
Table 148. Authors List of This Report
List of Figures
Figure 1. Gene Cell Therapy CDMO Picture
Figure 2. Global Gene Cell Therapy CDMO Market Size Comparison by Type (2020-2031) & (US$ Million)
Figure 3. Global Gene Cell Therapy CDMO Market Share by Type: 2024 VS 2031
Figure 4. Immune Cells Features
Figure 5. Stem Cell Features
Figure 6. Viral Vector Features
Figure 7. Plasmid DNA Features
Figure 8. Global Gene Cell Therapy CDMO Market Size Comparison by Stage (2020-2031) & (US$ Million)
Figure 9. Early-stage CDMO and preclinical CDMO Features
Figure 10. Clinical-stage CDMO Features
Figure 11. Commercial-stage CDMO Features
Figure 12. Global Gene Cell Therapy CDMO Market Size Comparison by Production Mode (2020-2031) & (US$ Million)
Figure 13. Centralized Factory Model Features
Figure 14. Distributed Regional Production Model Features
Figure 15. Global Gene Cell Therapy CDMO Market Size by Application (2020-2031) & (US$ Million)
Figure 16. Global Gene Cell Therapy CDMO Market Share by Application: 2024 VS 2031
Figure 17. Pharmaceutical and Biotechnology Companies Case Studies
Figure 18. Research and Academic Institutions Case Studies
Figure 19. Hospital Case Studies
Figure 20. Others Case Studies
Figure 21. Gene Cell Therapy CDMO Report Years Considered
Figure 22. Global Gene Cell Therapy CDMO Market Size (US$ Million), Year-over-Year: 2020-2031
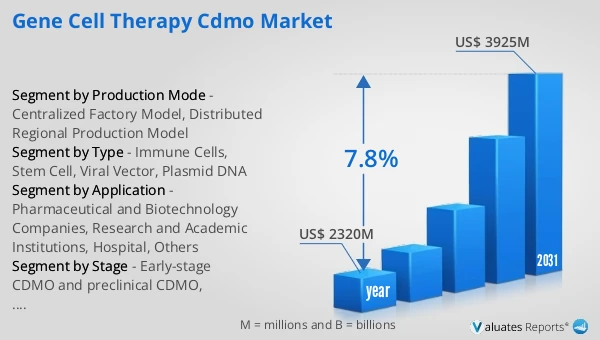
Figure 23. Global Gene Cell Therapy CDMO Market Size, (US$ Million), 2020 VS 2024 VS 2031
Figure 24. Global Gene Cell Therapy CDMO Market Share by Region: 2024 VS 2031
Figure 25. Global Gene Cell Therapy CDMO Market Share by Players in 2024
Figure 26. Global Gene Cell Therapy CDMO Market Share by Company Type (Tier 1, Tier 2, and Tier 3)
Figure 27. The Top 10 and 5 Players Market Share by Gene Cell Therapy CDMO Revenue in 2024
Figure 28. North America Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 29. North America Gene Cell Therapy CDMO Market Share by Country (2020-2031)
Figure 30. United States Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 31. Canada Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 32. Europe Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 33. Europe Gene Cell Therapy CDMO Market Share by Country (2020-2031)
Figure 34. Germany Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 35. France Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 36. U.K. Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 37. Italy Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 38. Russia Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 39. Ireland Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 40. Asia-Pacific Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 41. Asia-Pacific Gene Cell Therapy CDMO Market Share by Region (2020-2031)
Figure 42. China Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 43. Japan Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 44. South Korea Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 45. Southeast Asia Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 46. India Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 47. Australia & New Zealand Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 48. Latin America Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 49. Latin America Gene Cell Therapy CDMO Market Share by Country (2020-2031)
Figure 50. Mexico Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 51. Brazil Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 52. Middle East & Africa Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 53. Middle East & Africa Gene Cell Therapy CDMO Market Share by Country (2020-2031)
Figure 54. Israel Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 55. Saudi Arabia Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 56. UAE Gene Cell Therapy CDMO Market Size YoY Growth (2020-2031) & (US$ Million)
Figure 57. Catalent Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 58. Lonza Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 59. Charles River Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 60. Oxford Biomedica Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 61. Novartis Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 62. OBiO Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 63. GenScript Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 64. Pharmaron Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 65. Porton Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 66. Resilience Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 67. SK Pharmteco Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 68. Forge Biologics Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 69. Andelyn Biosciences Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 70. Roslin CT Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 71. eXmoor Pharma Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 72. Advanced Therapies Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 73. Catalent Biologics Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 74. Patheon Pharma Services Revenue Growth Rate in Gene Cell Therapy CDMO Business (2020-2025)
Figure 75. Bottom-up and Top-down Approaches for This Report
Figure 76. Data Triangulation
Figure 77. Key Executives Interviewed